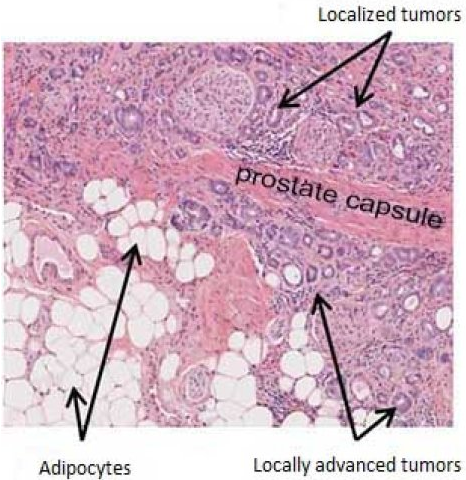
An experimental urine test can detect changes in prostate cancer-related genes, the test identified 92% of high-grade prostate cancer patients who are associated with elevated PSA levels. The study was published online in the JAMA Oncology. Click here to read the relevant literature.
 “This detection method can reduce the number of times each year hundreds of biopsies, reduce pain and risks associated with invasive diagnosis of the patient brings, this is not trivial.” The first author of the study, Dr. James McKiernan of Columbia University Medical Center ( PRESIDENT CUMC) of. In addition, the test is a urine-based detection technology, does not require digital rectal examination, the diagnosis of the environment there is no special requirements.
“This detection method can reduce the number of times each year hundreds of biopsies, reduce pain and risks associated with invasive diagnosis of the patient brings, this is not trivial.” The first author of the study, Dr. James McKiernan of Columbia University Medical Center ( PRESIDENT CUMC) of. In addition, the test is a urine-based detection technology, does not require digital rectal examination, the diagnosis of the environment there is no special requirements.
Although the PSA blood test in screening for prostate cancer is very common, but its clinical value has been questioned. PSA levels rise now, it is likely to be the only indicator of whether a patient is suffering from cancer, but they are not able to efficiently and reliably distinguish between high-level and low-grade prostate cancer. Need to make a distinction is due to prostate cancer patients in clinical high-level and low-level has a different treatment strategies: prostate cancer patients with low levels of the most common therapeutic strategy for active surveillance and watchful waiting, while high-grade prostate cancer patients need surgery or radiotherapy.
In such circumstances, PSA check high false positive rate has been much criticized. US Preventive Services Task Force (US Preventive Services Task Force) even opposed the application based on PSA screening tool, because only about 25% of the PSA is elevated in men with prostate cancer does change.
The only way that can be used to diagnose tissue biopsy, but this is a kind of high-invasive diagnostic methods and patient bring more suffering, is likely to cause hemorrhage and infection, even a very small part of the patient therefore and he died.
“Therefore, clinicians often recommend to patients with elevated PSA levels biopsy, even though most people probably do not have cancer.” Said Dr. McKiernan, “In other words, most people have absolutely no need for these invasive biopsy. ”
In this study, the researchers obtained 774 patients with prostate biopsy data, with their PSA level with another composite score to compare. The composite score to the urine test, based on the other risk factors contained additional PSA level, age, race and family history, including.
Researchers refer to 255 patients a score developed a cutoff score of 15.6 points, patients with a score greater than 15.6 then the patient is high-grade prostate cancer (equivalent to Gleason score 7 and above), patients with scores less than or equal 15.6 points is proof patients low-grade prostate cancer or illness.
Analysis shows that this fraction can be accurately identified 92% of high-grade prostate cancer. However, 66% of low-grade prostate cancer and healthy men, this test also evaluated as a high-grade prostate cancer. In clinical practice, the use of this test may enable the incidence of non-necessity of reducing prostate biopsies by 27%.
In 138 patients with low scores, 91% is really low-grade prostate cancer or healthy men, which means they do not have to be treated immediately. But we can also see a problem, “After the detection of the assessed, we found that patients with lower scores usually refuse to accept further biopsy.” Dr. McKiernan said.
This means that, for those with high-grade prostate cancer was diagnosed as error rates lower in patients who are likely to miss critical time for treatment. There are 12 people with high-grade prostate cancer but low ratings, nine of whom are suffering from aggressive prostate cancer risk, while three people with high-risk prostate cancer.
The test is called ExoDxTM Prostate (IntelliScore), for inspection and prostate cancer is closely related to the development process of ERG, PCA3 RNA and SPDEF gene. Based on the current study, the detection technology in distinguishing whether the patient has high-grade prostate cancer is superior to the current testing standards (AUROC 0.72 ratio 0.63; AUROC value of 1.0 is optimal; AUROC effect equivalent to a value of 0.5 on behalf of test random guess).
Prostate cancer is a common cancer, is the world’s second-largest of the four male tumors. For the diagnosis and treatment of prostate cancer, we also need more testing, commissioning more research.