Using microfluidic technology, scientists have developed a way to squeeze immune system B cells so their
membranes develop temporary holes, allowing the insertion of antigens that program specific immune
responses.
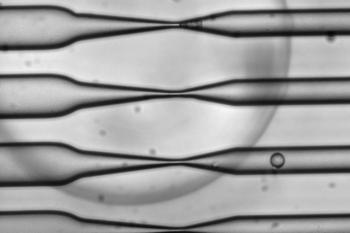
As cells squeeze through the narrowing channels, small temporary holes open in their membranes, allowing target antigens to enter before they close again.
Image credit: SQZ Biotech
The technique promises to open new ways to realize the dream of immunotherapy – using patients’ own immune
systems to fight disease. So far, the idea has proved hard to translate from lab
to clinic.
The researchers – from Massachusetts Institute of Technology (MIT) in Cambridge, MA – describe their work in
a paper published in the journal PLOS ONE.
There, they explain how their approach uses B cells to create “antigen-presenting cell vaccines” that could
train a patient’s immune system to fight disease.
An antigen – short for antibody generator – is any material that triggers an attack from the immune system.
This includes, for example, a protein from a disease-causing bacterium, or a piece of a virus particle.
Previous cell vaccine studies have tended to focus on dendritic cells as antigen-presenters. Dendritic cells
are versatile scouting cells that go around sampling and processing antigens and displaying the results on their
cell surfaces.
They travel to lymph nodes and the spleen, where they meet up with killer T cells that then use the presented
antigens to help them target and destroy the invading pathogens or cancer cells.
But the MIT team says there are drawbacks to using dendritic cells for cell-based vaccines. They have a short
lifespan, they do not divide once they are activated, and they are relatively sparse in the bloodstream.
The researchers were attracted to the idea of using B cells because they can proliferate when activated and
they are abundant in the bloodstream. But they are limited, in that unlike dendritic cells, they do not
constantly sample antigens – they are genetically programmed to process only antigens that match the receptor on
their cell surface.
How to get B cells to be as versatile as dendritic cells
Lead author Gregory Szeto, a postdoc at MIT’s Koch Institute for Integrative Cancer Research, explains the
challenge they faced in their study:
“We wanted to remove an important barrier in using B cells as an antigen-presenting cell population,
helping them complement or replace dendritic cells.”
The solution they describe in their paper is a mechanical one. Using a microfluidic device, they found they
could squeeze the B cells so they temporarily distort, opening their membranes just long enough to allow the
insertion of target antigens into the cells.
The device comprises tiny parallel channels etched on a microchip. The channels become progressively
narrower, and are designed to squeeze particles the size of a cell.
Using a positive pressure system, the researchers passed a suspension of B cells and target antigen through
the channels. As a cell squeezes through the narrow part of the channel, small, temporary holes open in its
membrane, allowing the target antigen to diffuse into the cell.
The antigen does not penetrate deep into the B cell, but remains trapped in the cytosol – within the
cell membrane – a result that has the same effect as presenting the target antigen.
Thus, sporting the target antigen, the B cells become cell vaccines equipped to prime the required response
from the killer T cells.
Proof of concept tested in mice
The researchers found that the squeezed and loaded B cells were as able to prime T cells as existing methods
that use antibody-coated beads.
They then inserted the squeezed B cells and antigen-specific T cells into mice and showed that the antigen-loaded B cells could expand T cells in the spleen and lymph nodes. This, in effect, proves the concept of using
B cells as a cell vaccine system, say the researchers.
The team also notes that theirs is the first method that effectively decouples antigen delivery from B-cell
activation, thereby conferring greater control over vaccine design.
Gail Bishop, a professor of microbiology at the University of Iowa Carver School of Medicine, was not involved
in the study. She says it presents a “creative new approach with considerable potential in the development of
antigen-presenting cell vaccines,” and adds:
“This new technical approach permits loading B cells effectively with virtually any antigen and
has the additional benefit of targeting the antigens to the CD8 T-cell presentation pathway, thus facilitating
the activation of the killer T cells desired in many clinical applications.”
The team now plans to refine the method to optimize distribution and function of the immune cells in the
body. They also envisage technology improvements that will result in a bedside or field-deployable device, with
the potential to offer cheaper and more effective cell-based therapies for patients.
Some of the authors declared a financial interest in SQZ Biotechnologies, the company that developed the microfluidic technology they used in the study.
Meanwhile, another MIT team concluded in a study recently described by Medical News Today that immunotherapy stands a
better chance of fighting cancer when it enlists both arms of the immune
system.
Writing in the journal Cancer Cell, Dane Wittrup, a professor in chemical engineering, and
colleagues describe how combining attacking tumors with antibodies (which enlists the innate immune response)
with adoptive T cell therapy (part of the adaptive immune response) successfully halted a very aggressive type
of melanoma in mice.