The immune cells in the human body are derived from hematopoietic stem cells in the bone marrow, and the morphology and function of various immune cells differentiated from hematopoietic stem cells are the result of cell differentiation.
Immune suppressor cells called MDSCs (myeloid-derived suppressor cells) may be important in developing treatments for advanced prostate cancer, according to a study at The University of Texas MD Anderson Cancer Center.
MDSCs are immune cells that “expand” when faced with cancer, inflammation or infections, giving them the unique ability to suppress the body’s T-cell response to disease.
“The tumor microenvironment is made up of a mixture of fibroblasts, infiltrating immune cells and other cells, proteins and signaling molecules,” said Y. Alan Wang, Ph.D., associate professor of Cancer Biology. “MDSCs are infiltrating immune cells that promote tumors through their striking lack of immunological response.”
Wang and fellow scientists Ronald A. DePinho, M.D., professor of Cancer Biology, and president of MD Anderson, Xin Lu, Ph.D., instructor, and Guocan Wang, Ph.D., postdoctoral fellow, both of Cancer Biology were co-authors on a paper about the study’s finding, which was published in the Dec. 20, 2015 online issue of Cancer Discovery.
Many previous studies have demonstrated a direct tie between MDSC and tumors, yet understanding of MDSC’s role in tumor progress, particularly in prostate cancer, has remained largely speculative.
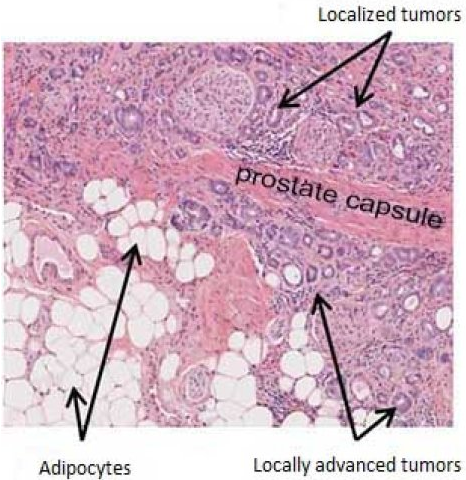
Using a novel prostate cancer mouse model and patient tumor samples, Wang’s and DePinho’s team showed that depletion of MDSCs suppresses tumor progression. In addition, they also revealed that a cell signaling pathway called Hippo-Yap1 regulated the protein Cxcl5 which was identified as a cancer-secreted chemokine that attracted or “recruited” MDSCs which expressed another protein, Cxcr2. The team was able to show that by blocking the Cxcr2 proteins with small molecule inhibitors, tumor progress was impeded. They also demonstrated that silencing of Yap1 expression in established tumors led to a reduction in MDSC infiltration and inhibition of tumor growth.
“Pharmacologic elimination of MDSCs or blockage of the Cxcl5-Cxcr2 signaling pathway resulted in robust anti-tumor response in vivo and prolonged survival,” said DePinho. “The targeting of either MDSC recruitment or infiltrated MDSCs may represent a valid therapeutic opportunity in treating advanced prostate cancer.”
Wang believes that further studies are needed, which may include combining MDSC inactivation with immunotherapy, including immune checkpoint inhibitors such as anti-CTLA4, anti PD1 and anti-PD-L1 antibodies.