The first steps toward developing “bioartificial” replacement limbs that are suitable for transplantation have reportedly been taken by a team at Massachusetts General Hospital in Boston, who write about their work with an animal model in the journal Biomaterials.
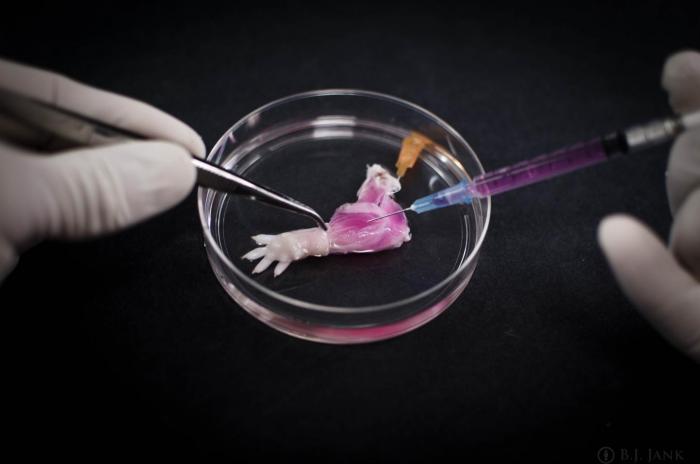
The researchers stripped away tissue from the rats’ forelimbs, but preserved the primary vasculature and nerve matrix.
Image credit: Dr. Bernhard Jank/Ott Laboratory
More than 1.5 million Americans have lost a limb. Prosthetic limbs have greatly advanced in function and appearance, but the authors of the new paper acknowledge that the devices still have many limitations.
Some patients over the past 20 years have received hand transplants from donors, but this procedure comes attached with lifelong risks from immunosuppressive therapy.
This problem could be solved by using the patient’s own progenitor cells to regenerate the tissue for a new limb – rather than rely on a donor – but an appropriate matrix or scaffold has yet to be devised on which scientists would be able to grow the new tissue.
“The composite nature of our limbs makes building a functional biological replacement particularly challenging,” explains senior author Dr. Harald Ott, of the Massachusetts General Hospital Department of Surgery and the Center for Regenerative Medicine.
Dr. Ott continues:
“Limbs contain muscles, bone, cartilage, blood vessels, tendons, ligaments and nerves – each of which has to be rebuilt and requires a specific supporting structure called the matrix. We have shown that we can maintain the matrix of all of these tissues in their natural relationships to each other, that we can culture the entire construct over prolonged periods of time, and that we can repopulate the vascular system and musculature.”
In animal models, Dr. Ott and colleagues have previously been able to regenerate kidneys, livers, hearts and lungs, using a detergent solution to strip living cells from the donor organ, which is then repopulated with appropriate progenitor cells.
However, the new study represents the first use of this technique to engineer a bioartificial limb, which is more complex.
Cellular material was stripped from the limbs of dead rats, but nerve matrix was preserved
In deceased rats, the researchers stripped away the cellular material from the animals’ forelimbs over the course of 1 week, but preserved the primary vasculature and nerve matrix. This remaining material provided a structure for all of the composite tissues required by the limb.
This forelimb matrix was then populated by cultures of muscle and vascular cells in a bioreactor. The vascular cells were injected into the main artery of the limb, in an effort to regenerate veins and arteries. The progenitor cells, meanwhile, were injected into sheaths in the matrix that define muscle positions.
The limb was electrically stimulated after 5 days to encourage muscle formation. After 2 weeks, the limb was removed from the bioreactor. The researchers found that the electrical stimulation caused the new muscle fibers to contract with a strength that was 80% that of a newborn rat.
When the forelimbs were transplanted into recipient rats, blood quickly began to circulate in the new limb, and when the muscles within the graft were stimulated electrically, the wrists and digital joints of the rats’ paws flexed appropriately.
The team also decellularized baboon forearms successfully, which the authors claim confirms the feasibility of using the technique on a scale similar to human patients.
However, the researchers still face the challenge of reintegrating the regrown nerves of a regenerated limb into the recipient’s nervous system.
“In clinical limb transplantation, nerves do grow back into the graft, enabling both motion and sensation, and we have learned that this process is largely guided by the nerve matrix within the graft,” Dr. Ott says.
Dr. Ott hopes that the same logic will apply to bioartificial grafts. Next, the team will attempt muscle regeneration using human cells, before expanding the process to human bone, cartilage and connective tissue.
Written by David McNamee