One of the hallmarks of Alzheimer’s disease is deposits of beta-amyloid – a
faulty protein – in the brain. Surrounding these deposits or plaques are abnormally
high numbers of lysosomes, tiny specialized systems that digest and dispose of
cellular garbage.
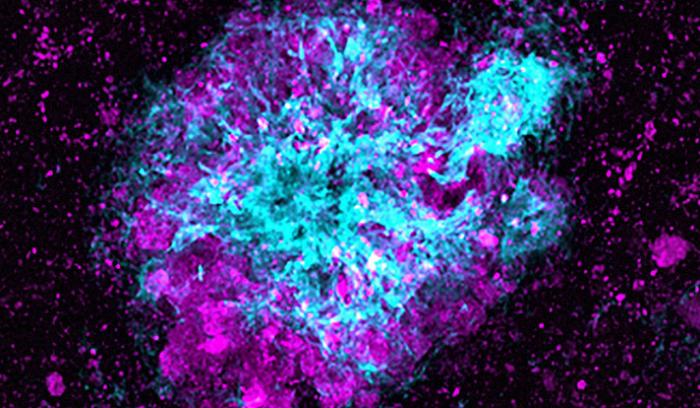
Lysosomes – in magenta – accumulate near amyloid plaques.
Alzheimer’s disease is the most common form of dementia. It affects the part of
the brain that controls thought, memory and language.
The disease begins with mild memory loss and progresses to the point where the
person affected struggles to carry on a conversation and respond to what is
happening around them.
According to the Centers for Disease Control and Prevention, in 2013, as many as
5 million Americans aged 65 years or older had Alzheimer’s disease, and the number
is expected to rise to 13.8 million by 2050.
When scientists first found – around 50 years ago – that beta-amyloid deposits
in the brains of people with Alzheimer’s disease were surrounded by massive
accumulations of lysosomes, they assumed they were there to help degrade and clear
away the faulty protein.
Now, a new study from Yale University of New Haven, CT, suggests not only is it
not as straightforward as this, but the opposite could be happening.
The Yale team found that the lysosomes surrounding the deposits of
beta-amyloid are faulty, and instead of working against Alzheimer’s progression,
may even be contributing to it.
The researchers report their findings in the Proceedings of the National
Academy of Sciences.
They suggest the discovery could lead to new types of treatment that correct the
faulty lysosomes so they can do their job properly, clear away the beta-amyloid
and stop it clogging the brain.
Vicious cycle
In the study paper, the Yale team describes how the lysosomes build up in swollen axons or nerve
fibers that contact the amyloid deposits outside the brain cells.
They found that the lysosomes have abnormally high levels of beta-secretase, the
enzyme that triggers the production of the toxic beta-amyloid protein.
The researchers suggest the faulty lysosomes are not able to break down the
enzyme because they are not fully mature. To become mature, they have to travel
along the axons. But the beta-amyloid plaques block their ability to do this, which
results in more accumulation of beta-secretase, which in turn triggers more beta-amyloid.
First author Swetha Gowrishankar, a postdoctoral scientist in the lab of senior
author Shawn Ferguson, assistant professor of cell biology, sums up the
finding:
“We think this represents a vicious circle.”
Put simply, to be able to do their job of eliminating the enzyme that boosts
Alzheimer’s plaques, the lysosomes have to be able to travel and mature. But their
means to do this is blocked by Alzheimer’s plaques.
The team now plans to test in mice engineered to develop Alzheimer’s whether
tweaking genes to help the faulty lysosomes digest the beta-secretase protects
against disease progression.
Faulty lysosomes have also been implicated in other neurodegenerative
diseases including Parkinson’s and frontotemporal dementia, they note.
Funding for the study came from the Howard Hughes Medical Institute, the Ellison
Medical Foundation, the National Institutes of Health, and the Consortium for
Frontotemporal Dementia Research.
Meanwhile, Medical News Today recently learned how high blood sugar may contribute to Alzheimer’s
disease by speeding up the production of beta-amyloid. A study conducted in mice
found that a doubling of blood glucose led to 20% higher levels of the protein.
Written by Catharine Paddock PhD